|
The first thing we did was to make the sodium acetate by mixing baking soda and vinegar and the liquid that we had left over is what we call sodium acetate. The next thing we did is heat it up for a long time ‐ How did your experiment turn out? Did it work? To what extent? My expierement turned out great. It worked even better that I had planned. The crystals in the end formed faster than I imagined they would and most of all I think it looked really cool. ‐ Describe how your experiment looked. Do you have any images or can you illustrate artifacts? Yes I have a video ive included it below and it shows the last part of the expirement when we crystalized it by touching it with antother crystal so that it would grow off of that one and then it would make many crystals. -Did you get desirable results? If yes, what did you think you did that contributed to desirable results? If not, then what do you think contributed to undesirable results? To get the results we got, I beleive that we followed the instructions precisley so our project outcome was what we expected, example: In the end when we touched the crystals to the liquid sodium acetate all the crystals started to form and expand making the entire thing turn solid right before our eyes. How do you think you performed in this activity? I think that I preformed pretty well during this activity but like always I could've been better. I left while the crystals were on the hot plate and came back when it was done but I shouldve stayed the whole time just in case something happened. Who did you work with? I worked with Alexander Uppenkamp How do you think your group performed? Our group preformed pretty well because we performed and finished the expirement successfully. Like everything else we could have done better. The first attempt we had problems and had to try again the next day. If you could do this again, what would you change to get better results? If we would do this again we would stay in the room while we had it on the heat plate so if anything happened we could fix it right away. If you were organizing a group for the next lab activity, who would you choose to work with again? We would work together again because we have a background together wich means we understand how eachother works and were able to strengthen eachothers weakness' Do you see any way to continue this experiment? Would you want to? We could heat it up again and repeat the same process over and over again as many times you want. I actually am thinking about doing this because it looks so awesome when you crystalize it What skills, techniques, or pieces or wisdom did you get from this activity? We learned how to crystalize sodium acetate from scratch (only using baking soda and vinegar) What do you think happened on a molecular level? (Looking beyond) Once you have your crystalized sodium acetate you melt it down so it no longer has its solid molecule structure but as soon as you touch the liquid with a small crystal they begin to grow off of it and continue to grow until its all a solid. Video: https://drive.google.com/a/hightechhigh.org/file/d/0B6L47adaZfHySy02RFFiQnZnam8/view?usp=sharing
0 Comments
In Chemistry class the other day we decided to make disapearing ink. The first step was to measure out 0.1 grams of phenolphthalein into a flask then we added 10 mL of ethanol and mixed it up so it could dissolve. Then we added 100 mL of water and it turned into a milky white color and then we added 3 drops of sodium hydroxide and the entire thing turned dark red. The ethanol is there to help in the distillation of the water. As the the ink is exposed is to carbon and the air the pH level drops because of the hydrogen ions that were made in the mixture, that makes the ink turn from red to clear.
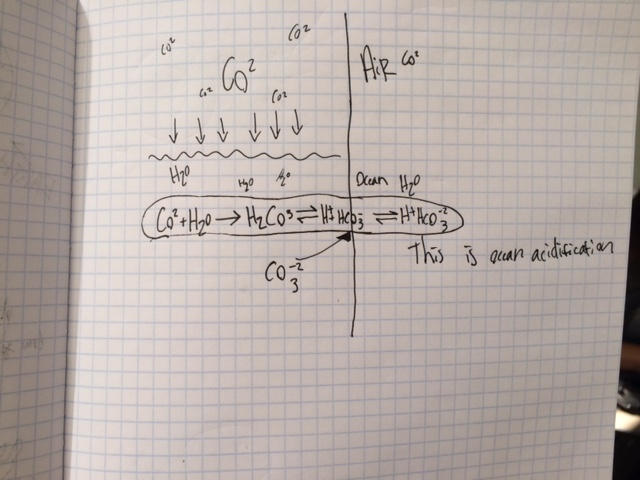
As we know human activity creates CO2 and puts it into our atmosphere. When the CO2 is dispersed 30%-40% of it is put into our ocean the rest either ends up staying in the atmosphere or on land helping plants or something. All that carbon dioxide creates the oceans acidity to rise. The CO2 in the water reacts with the water (H20) creating carbonic acid. If you look at the equation below, the process of all this makes it so all the hydrogen ions are freed and are left floating around which is what causes the acidity to rise. And this is pretty normal for the acidity to rise but the rate that its changing is much faster than anything that we've seen in a long time. And when the pH is lowering its causing coral to be destroyed and its disturbing animal life such as fish and crustaceans. So I believe that If we stop using so many fossil fuels and that if we try our best to not drive or waste electricity we can slow down this process of destroying our oceans.
The one thing that kept reoccurring in this video is when Neil deGrasse Tyson answers any question he is very careful and wise with his words and although he is a very smart we'll educated man he answers by saying my answer is “ ” but that's my answer someone else could be thinking something entirely different. But he could just take his word for fact since that's what us humans do, we take in information which could be true or false and depending on our knowledge of that person we say “Yeah right like he knows what he's talking about.” Or we could say this is a teacher this must be right. But Neil doesn't tell us what's right he suggests something and lets us ponder that and decide for ourselves. Also another thing that I really found inspiring was when he said that we thought our planet was the only one, then we found out about our whole solar system. We thought our sun was special, then we found billions just like it, then our galaxy and then he say that there could be other universes like ours but slightly different and that we need to open ourselves up to all the different possibilities that are out there. I think the main thing that he’s trying to get across is that our world is vast and we will continue to explore and that we will never stop progressing.
In Andrews class the other day we learned about properties of matter such as Pressure Buoyancy and Density. All three of these things are vitally important because say you have a trash bag. just a regular old trash bag but, if how decrease the density by raising the temperature it will float away! We did this in class by making floating candles it was fun and a good way to understand the properties of matter.
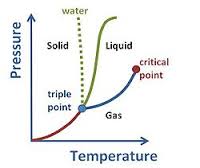
Density is the amount of mass per volume so mass/volume. So to change the density of something you should change the ratio, up or down depending on what your trying to do. Buoyancy is how much something will float wether its gas or a liquid. Buoyancy is determined mostly on the density of an object. Pressure is the sum of collisions of gas particles on the walls of a container. There are three properties of matter Solid, Liquid and gas. Some things that can change an objects physical form are pressure decrease/increase and the raising or lowering of temperature. I have included a chart that helps show this. It helped me understand what and how all of this works. As you can see from the chart any state of mater can skip to another which was one thing I did not know until our dry ice experiment. I learned that a solid can actually go straight to a gas! I also learned that this process is called deposition.
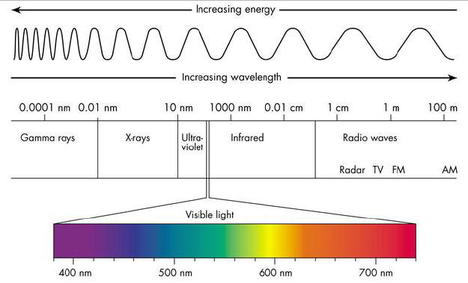
Below are the names of the process of matter changing its form from each specific state into another. Gas^Liquid= Condensation Liquid^Gas= Vaporization Liquid^Solid= Freezing Solid^Liquid= Melting Solid^Gas= Sublimation Gas^Solid= Deposition Light is energy made up from wavelengths that make it so we can see them, our eyes only let us see so much, there are so many different sizes wavelengths and we only are able to see a tiny bit of all that. Those different types of wavelengths that I mentioned earlier are defined by the amount of energy they have such as Gamma rays, X-rays, Ultraviolet rays, Infrared, and one that I personally enjoy listening is, Radio Waves. To make it easier to comprehend I included a diagram.
Combustion is basically the process of burning something. For combustion to take place there must be three key components: A fuel, an oxidizer and something to ignite the fuel. The ignition will light the fuel then the oxidizer feeds the burning fuel oxygen which makes the fire bigger. Fuels can be in many different forms they can be in the form of solids, liquids or gas. Something we did during class to get a better understanding was we created hydrogen by using magnesium and citric acid and it filled up a balloon then we burned it. Personally I'm a hands on learner so this was great for me. When we burned our balloons I believe that the candle ignited our hydrogen and then since it was a gas all the oxygen around it oxidized it and created an explosion (Look at the image below) Update: On tuesday we actually went into great detail on what exactly happened on an atomic level. When we mixed magnesium with the citric acid we created hydrogen. When we added heat to the whole equation all the atoms started to move around and that released energy which is what we saw as an explosion.
How would describe what science is? I believe that science can give us all the information that we need to explain what everything is, where it comes from, how stuff works. Science not only can tell you all about the Human race but about animals, chemicals and even our very own planet earth. Science can tell you everything you need to know about the world around you. That, is science. What is a hypothesis and why would you make one?
A hypothesis is an educated guess. Before you do an experiment you should make a hypothesis on what you think will happen during and after. You make these because its good to see what works and what doesn't and then you can improve your experiments. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2016
Categories |

 RSS Feed
RSS Feed
